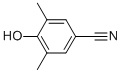
3,5-Dimethyl-4-hydroxybenzonitrile CAS#: 4198-90-7; ChemWhat Code: 62810
Identification
| Renseignements sur les brevets | ||
| ID de brevet | Titre | Date de publication |
| WO2023 / 176725 | ALLYL ETHER COMPOUND AND METHOD FOR PRODUCING SAME, CURABLE RESIN COMPOSITION, VARNISH, PREPREG, CURED PRODUCT, POLYPHENYLENE ETHER RESIN CURING AGENT, AND CRYSTALS AND METHOD FOR PRODUCING SAME | 2023 |
| CN104530078 | A thieno [3, 2 – d] pyrimidine derivative and its preparation method and application | 2017 |
| US2013 / 23563 | AMIDINOANILINE DERIVATIVE | 2013 |
| EP2213650 | SUBSTITUTED DIPHENYLAMINES AS INHIBITORS OF REVERSE TRANSCRIPTASE, PROCESS OF PREPARING THEM AND USE THEREOF | 2010 |
Données physiques
| Aspect | Blanc solide |
| Point de fusion, ° C | Solvant (point de fusion) |
| 72 – 74 | |
| 165 – 166 | |
| 96 – 98 | |
| 125 | aq. éthanol |
| 124 – 125 | |
| 124 – 124.4 | éther de pétrole |
Spectra
| Description (spectroscopie RMN) | Noyau (spectroscopie RMN) | Solvants (spectroscopie RMN) | Fréquence (spectroscopie RMN), MHz |
| Déplacements chimiques, spectre | 1H | chloroforme-d1 | 400 |
| Déplacements chimiques, spectre | 1H | chloroforme-d1 | |
| Déplacements chimiques, spectre | 13C | chloroforme-d1 | |
| Déplacements chimiques, spectre | 1H | diméthylsulfoxyde-d6 | 400 |
Route de synthèse (ROS)
| Conditions | Rendement |
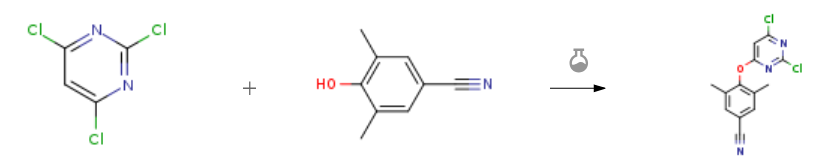
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 70℃; for 2h; Procédure expérimentale A mixture of 2,4,6-trimethylpyrimidine II(110 mmol, 20.0 g) and 3,5-dimethyl-4-hydroxybenzonitrile III (110mmol, 16.2 g) Was reacted with N, N-diisopropylethylamine (DIEA) (132 mmol, 17. Og) in 100 mL of 1,4-dioxane at 70 ° C for 2 h, When the reaction solution was cooled to about 10 ° C, A 200 mL aqueous solution was slowly added to the reaction solution and stirred for 30 min, filtered and dried in vacuo to give 29.8 g of intermediate IV in a yield of 92.5% | 92.5% |
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 70℃; for 2h; Procédure expérimentale 4.1.4 4-((2,6-Dichloropyrimidin-4-yl)oxy)-3,5-dimethylbenzonitrile (7) 2,4,6-trichloropyrimidine (6, 11mmol, 2.0g), DIEPA (13mmol, 1.7g) and 4-hydroxy-3,5-dimethylbenzonitrile (11mmol, 1.6g) were dissolved in 10mL 1,4-dioxane and the mixed solution were heated at 70°C for 2h. After the reaction mixture was brought to room temperature, 50mL cold water was poured into the mixture and stirred for another 30min, filtrated. The wet cake was dried at 55-60°C under vacuum to give the intermediate 7 as white solid with a yield of 92%, mp: 207-209°C. 1H NMR (400MHz, DMSO-d6, ppm) δ: 7.76 (s, 2H, C3,C5-Ph-H), 7.64 (s, 1H, pyrimidine-H), 2.12 (s, 6H). ESI-MS: m/z 294.2 [M+1]+. C13H9Cl2N3O (293.01). | 92% |
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 70℃; for 2h; Procédure expérimentale Weigh 2,4,6-trichloropyrimidine (2.0g, 10.9mmol), 4-hydroxy-3,5-dimethylbenzonitrile (1.6g, 10.9mmol) and N,-diisopropylethylamine (3.6ml, 21.8mmol) in 25mL of 1,4-dioxane solution, stirred at 70 °C for 2h. After the reaction was detected by TLC, after the reaction solution was cooled, 100 mL of water was slowly added thereto, stirring was continued for 30 min, filtration was performed, and the vacuum drying oven was used for drying. A white solid was obtained as the compound 4-((2,6-dichloropyrimidin-4-yl)oxy)-3,5-dimethylbenzonitrile (2) in a yield of 91.8%. | 91.8% |
Sécurité et dangers
| Pictogramme (s) |   |
| Signal | danger |
| Mentions de danger SGH | H300 (86.67%) : Mortel en cas d'ingestion [Danger Toxicité aiguë, orale] H302 (11.11%): Nocif en cas d'ingestion [Avertissement Toxicité aiguë, par voie orale] H315 (13.33%): Provoque une irritation cutanée [Avertissement Corrosion cutanée / irritation cutanée] H319 (100%): Provoque une sévère irritation des yeux [Attention: Lésions oculaires graves / irritation oculaire] H335 (11.11%): Peut irriter les voies respiratoires [Attention Toxicité spécifique pour certains organes cibles, exposition unique; Irritation des voies respiratoires] |
| Codes de déclaration de précaution | P261, P264, P264+P265, P270, P271, P280, P301+P316, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501 (L’énoncé correspondant à chaque code P est disponible à l’adresse suivante: Classification SGH page.) |
Autre informations
| À la température ambiante et à l'abri de la lumière | |
| code SH | |
| Stockage | À la température ambiante et à l'abri de la lumière |
| Field Intelligence | 1 année |
| Prix du marché |
| Ressemblance à la drogue | |
| Composant des règles Lipinski | |
| Masse moléculaire | 94147.177 |
| logP | 1.791 |
| HBA | 1 |
| HBD | 1 |
| Règles Lipinski correspondantes | 4 |
| Composant de règles Veber | |
| Surface polaire (PSA) | 44.02 |
| Lien rotatif (RotB) | 0 |
| Règles Veber correspondantes | 2 |
| Motif d'utilisation |
| 3,5-Dimethyl-4-hydroxybenzonitrile CAS 4198-90-7 used as an intermediate in the synthesis of various drugs. |
Acheter un réactif | |
| Pas de fournisseur de réactifs? | Envoyez une demande rapide à ChemWhat |
| Vous voulez être répertorié ici en tant que fournisseur de réactifs? (Service payant) | Cliquez ici pour contacter ChemWhat |
Fabricants approuvés | |
| Vous souhaitez être répertorié comme fabricant approuvé (approbation requise) ? | Veuillez télécharger et remplir ce document et renvoyer à [email protected] |
Contactez-nous pour une autre aide | |
| Contactez-nous pour d'autres informations ou services | Cliquez ici pour contacter ChemWhat |